Bespoke Participant Information Leaflet (PIL).
Optimising the participant information leaflet (PIL) through a particular, bespoke process involving formal user testing makes little or no difference to recruitment.
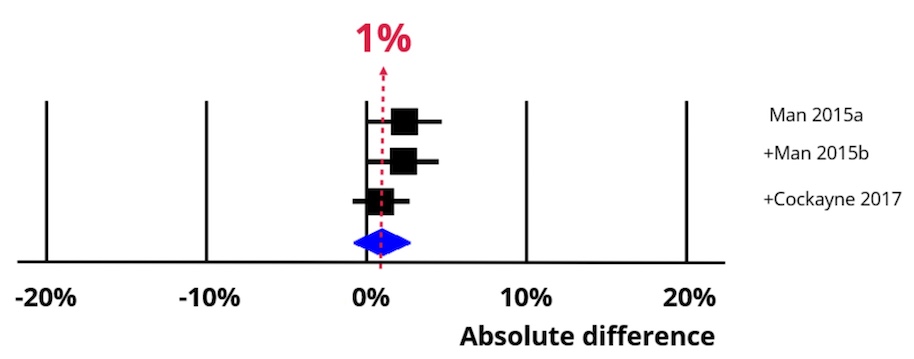
An increase of 1% (95% confidence interval = -1% to 3%).
GRADE High certainty.
We recommend against using this intervention with the intention of increasing recruitment. Evaluation outside the UK would be useful.
See Resource bundle below for details of how to use bespoke Participant Information Leaflets (PIL) or build a SWAT evaluation of them into your trial.
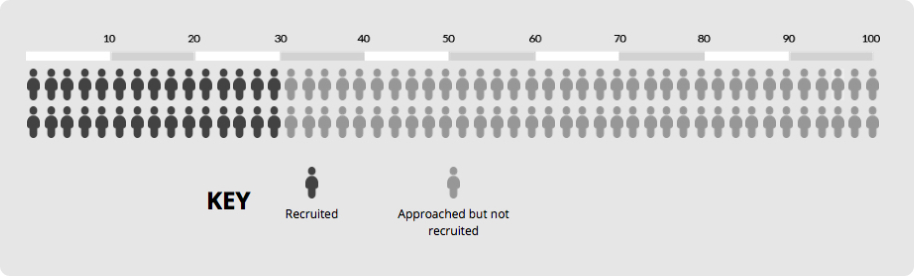
Imagine a trial that needs to recruit 30 participants and initial recruitment is 30% of those approached. This means you’d need to approach 100 people to recruit 30 of them (see chart).
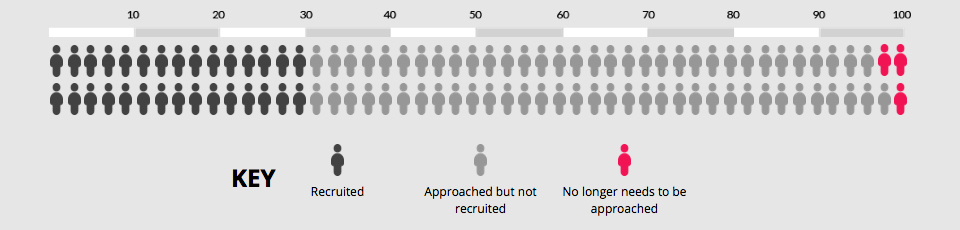
Now imagine using a bespoke user-centred PIL development process. The chart below shows the impact of an absolute increase of 1% (95% CI = 1-% to 3%). Recruitment is now 31%, which means our best estimate is that 97 people would now need to be approached to recruit 30 of them.
Trial Forge will make trials more efficient by looking for marginal gains across all trial processes, from research question to implementation into routine care. It will encourage everyone connected with trials to be more sceptical of what we do by asking for the evidence behind all of our trial decisions.