PRO EDI: improving how equity, diversity and inclusion are handled in trials and evidence synthesis
Equity, diversity and inclusion in trials and evidence synthesis
Few single randomised trials change practice, and this is how it should be. Instead, bodies of evidence from several trials are collated through evidence synthesis, generally systematic reviews. It is the results of these reviews that lead to practice and guideline changes. Because evidence synthesis can have important consequences for which care is provided, and how, it is essential that equity, diversity and inclusion is explicitly considered within synthesis.
PRO EDI is an initiative led by Trial Forge, Evidence Synthesis Ireland and the Campbell Collaboration to try and make it easier for those doing systematic reviews, especially ones including randomised trials, to explicitly consider equity, diversity and inclusion in their reviews. The initiative benefits from an international advisory group and from public involvement throughout its development. We think this helps to make PRO EDI relevant to all reviews and reviews, wherever the reviewers may be based in the world, and whatever the topic of the review. Our starting points were PROGRESS-PLUS, which maps out equity, diversity and inclusion topics that reviewers should consider; and the INCLUDE Ethnicity Framework, which has operationalised consideration of ethnicity for trial design.
Why is thinking about equity, diversity and inclusion important?
Here is a collection of over 20 examples of how health outcomes vary by characteristics such as age, gender, ethnicity and disability. It is the potential for these sorts of difference that make it so important to collect and report information on the PRO EDI characteristics.
What is PRO EDI?
Here is a 2.5 minute video explaining what PRO EDI is all about. The video was developed together with ScienceSplained, a science communication company.
Get hold of the PRO EDI paper
You can read more about how PRO EDI was developed in our paper in Cochrane Evidence Synthesis and Methods.
News: PRO EDI mandated at Trials!
The journal Trials will be mandating use of PRO EDI for trial protocols and reports of trial results submitted to the journal from 1st Jan 2026. The details are given in an editorial published on 9th Sept 2025.
How you can help
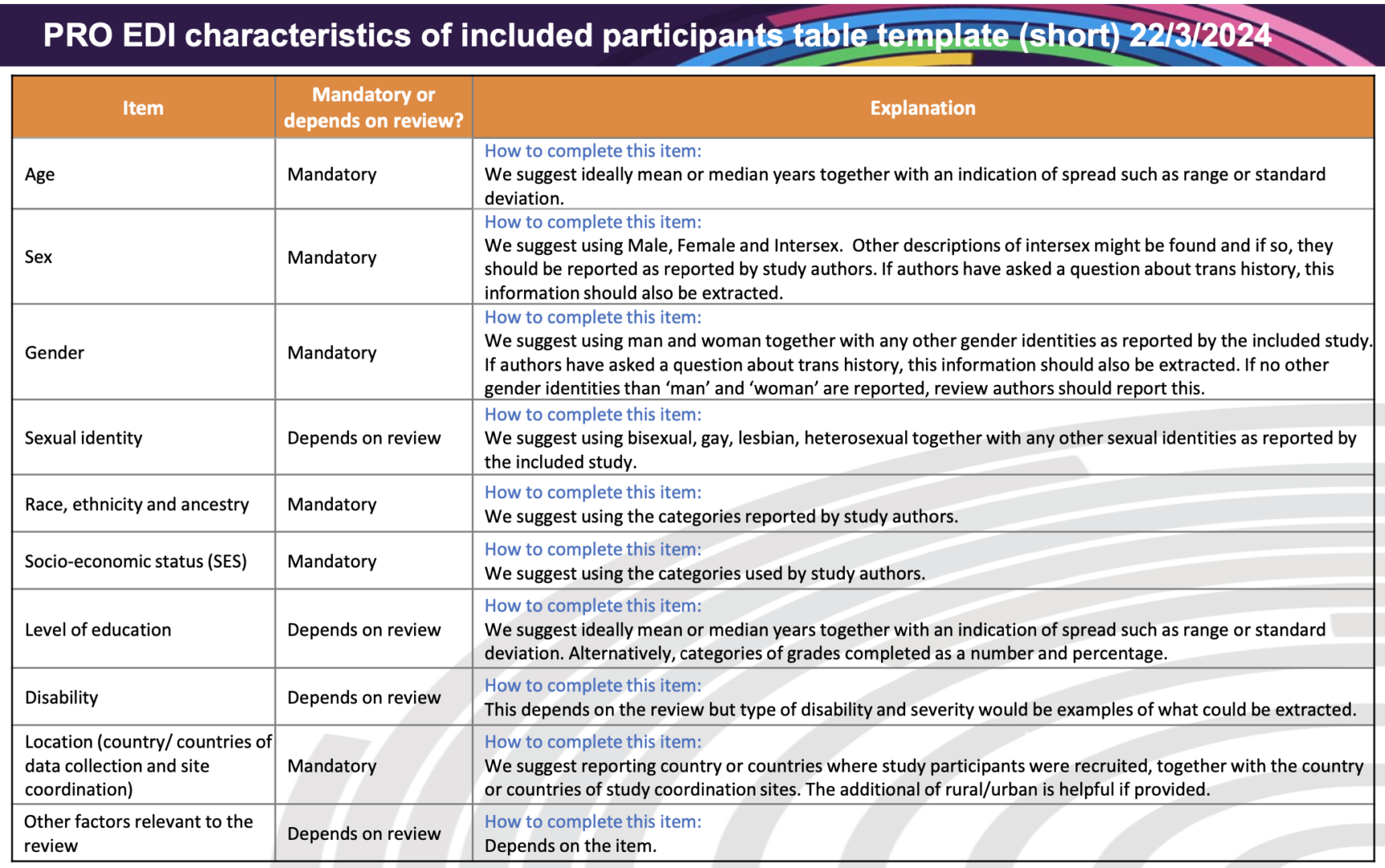
PRO EDI has developed a table for extracting information about participant characteristics from randomised trials included in a systematic review. Having these data is the starting point of being able to make judgements about whether the review findings apply equally to all those who could benefit from the treatment or initiative being reviewed.
We would be delighted if you used PRO EDI in your review. We would also be extremely grateful for examples you could send us (e.g., a link to a review) of PRO EDI being used. We would be interested in your general feedback too, especially on the time taken to use PRO EDI (e.g., does PRO EDI use up a lot of time, or more or less what you would have used before? Does it use less time?).
Note that currently, PRO EDI is only specifically designed for randomised controlled trials; however, we think it can be useful for other study designs too.
- The short version of the PRO EDI participant characteristics table (same items but less guidance) can be downloaded free here (pdf) and here (Word). An accessible version can be downloaded free here.
- The full version of the PRO EDI participant characteristics table can be downloaded free here (pdf), here (Word) and here (Powerpoint).
- We’ve designed a feedback form for you to use and are looking for one completed form per review, which can be downloaded here.
There is guidance in the PRO EDI table itself, but we also have guidance on how to use the information extracted using the PRO EDI table. This includes some examples although we aim to add more examples during 2025.
Any comments or questions about PRO EDI, please email info@trialforge.org. The Evidence for Policy and Practice Centre in the UK has also developed guidance on considering and reporting methods for investigating health equity issues in systematic reviews and other evidence syntheses, which includes links to PRO EDI.
Finally, some of the PRO EDI team gave a seminar for Evidence Synthesis Ireland on 17th October 2024 that presented the tool, and why it is important from a patient and public perspective. You can watch the seminar below, and there’s more information about the speakers here.
How to cite
PRO EDI participant characteristics table
PRO EDI participant characteristics table 22/3/2024; https://www.trialforge.org/trial-diversity/pro-edi-improving-how-equity-diversity-and-inclusion-is-handled-in-evidence-synthesis/ [insert access date].
Guidance
PRO EDI interpretation guidance 25/4/2024; https://www.trialforge.org/trial-diversity/pro-edi-improving-how-equity-diversity-and-inclusion-is-handled-in-evidence-synthesis/ [insert access date].
Licencing and permission to use
Once we have completed development work, the plan is to make the PRO EDI materials available for free here. You will not need a licence or to ask us for permission to use the PRO EDI table or associated documents. We only ask that you acknowledge the tool, if you have used it, by citing it (see citation on this page).
Funder