Making trials more informative – learn more
Learning about the 12 Good Practice Actions
The INFORM project identified 12 Good Practice Actions that were identified as key to producing informative trials. The following training modules give background context to each of the actions, and at the foot of each module, we suggest straightforward ways to help funders operationalise these.
Complying with our Good Practice Actions may have resource implications. For example, working with communities to develop effective and inclusive recruitment and retention strategies (Good Practice Action 3) requires appropriate funding. However, not all actions involve additional costs. Using structured tools such as those suggested at the design stage (Good Practice Action 4) is unlikely to require additional resources; trial teams simply need to use them. Where additional funding is needed, funders should consider providing it to ensure a trial is informative. It is important to recognise that lack of funding alone does not explain the prevalence of uninformative trials. Lack of awareness, insufficient methodological expertise within the trial teams, and the absence of structures to support or enforce actions such as those we propose are likely to be at least as important.
Links to existing resources are given and we also include a template grant form that funders can use to improve the likely informativeness of the trials they review and fund. This is referred to as the INFORM Structured Grant Form for Trials and is set out with questions and guidance for applicants. We believe this form can help funders to shape their pre-submission process to better align with producing informative trials.
We also have a short paper describing how we developed the 12 Good Practice Actions (Prowse et al, 2026).
The 12 Good Practice Actions
- Action 1:
- Action 2:
- Action 3: Engage patients and communities from the outset to build trust, ensure relevance, and strengthen recruitment and retention
- Action 4: Apply structured design tools, established frameworks, and Open Science practices (e.g., COMET, SPIRIT, pre-registration) to improve trial quality, transparency, and reproducibility
- Action 5:
- Action 6:
- Action 7:
- Action 8:
- Action 9:
- Action 10:
- Action 11:
- Action 12:
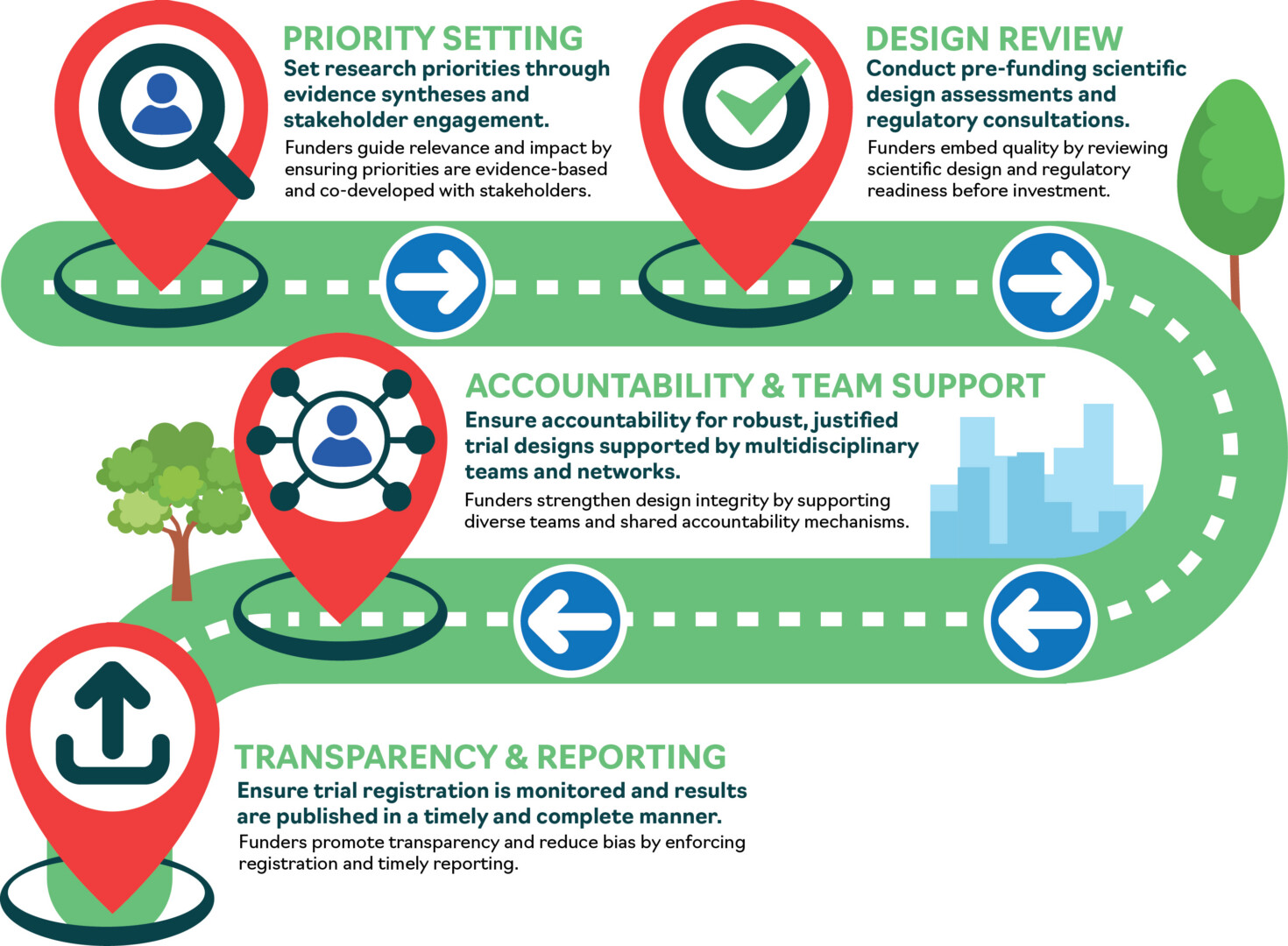
Figure 1 The INFORM Good Practice Actions pathway for funders: from priority setting through to transparent reporting. Pathways for other organisations are available in Resources.