Good Practice Action 7: Use future-proofing measures
Use future-proofing measures to safeguard trial integrity and ensure technologies remain relevant, reliable, and unbiased
Future-proofing involves anticipating changes in technology, healthcare context and research practice to maintain relevance, data integrity, minimise bias, and ensure that trial processes remain sustainable over time. Anticipating changes allows the trial team to make plans that safeguard the relevance and integrity of the trial and avoid research waste and exposure of participants to unnecessary risks.
Watch the video: INFORM Good Practice Action 7
Frances Shiely, Full Professor of Clinical Trials, University College Cork, Ireland explains why future-proofing your trial while designing it is essential to support an informative trial.
Future-proofing isn’t about predicting the future. It’s about building enough flexibility and foresight into your design that your trial remains rigorous, relevant, and unbiased, whatever changes come. Future-proofing is a planning activity, not a rescue operation.”
Trial teams and others should:
Technologies and practices might change during the trial. There may be shifts in the intervention, comparator, or methods during the study due to e.g. changes in technology, standard of care, or societal practice. The healthcare context itself may change, for example through the introduction of new government initiatives or policy.
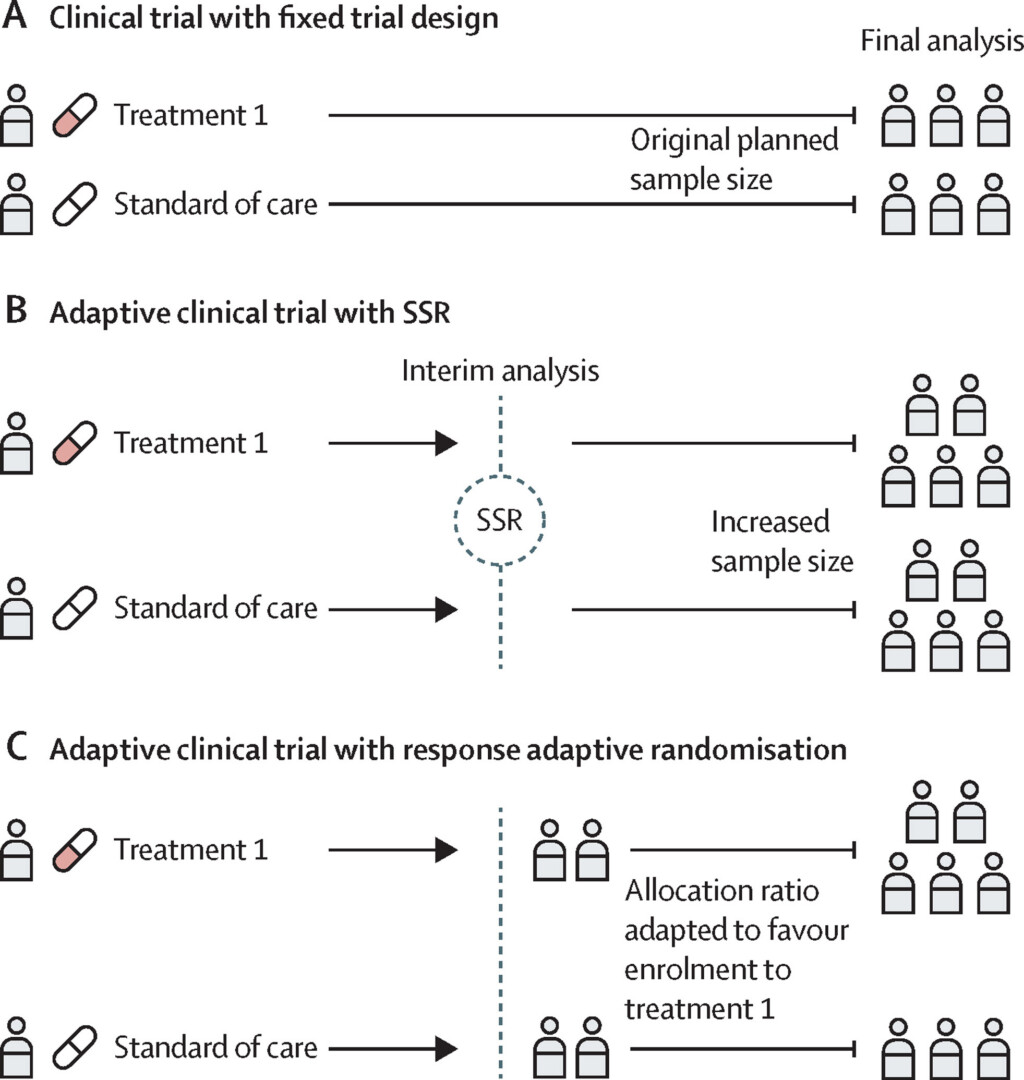
Adaptive trials are a way to run trials which are planned for flexibility, allowing modification of the trial in accordance with pre-defined rules (Pallmann et al., 2018). Research teams should consider the pros and cons of multiple design options (see Figure X), ideally with the use of statistical simulations to compare the efficiencies of different designs (Park et al., 2021).
Figure 1. Conventional fixed trial designs and common adaptive trial designs. (A) A two-arm randomised clinical trial with conventional fixed trial design. (B) A two-arm trial with sample size reassessment (SSR). If the first interim analysis shows worse results than expected, an SSR can be carried out by use of the interim results. An SSR is not permitted in a traditional non-adaptive trial, so even when the original planned sample size is reached, the trial might be underpowered. If SSR is permitted, the sample size could be increased to ensure that the trial is adequately powered. (C) A two-arm trial with response adaptive randomisation. The response adaptive randomisation design allows for preferential assignment of interventions that show favourable interim results. In this example, the response adaptive randomisation design allows for an increased allocation ratio to treatment 1 based on the interim results. From Park et al., 2021 licenced by CC BY 4.0.
An example of a flexible, adaptive approach is the Multi-Arm Multi-Stage (MAMS) trial design. This allows researchers to test multiple treatments, add new treatments as they become available, and stop treatments that show no promise. An example of a MAMS trial in practice is the STAMPEDE trial for prostate cancer.
Trial adaptability in the face of changing external factors is demonstrated by the next example. During the COVID-19 pandemic, an existing trial for young people with chronic pain pivoted to virtual assessment and treatment when in-person clinical research was unexpectedly halted. The authors report that this increased enrolment rates, and their data demonstrated acceptability, feasibility, and equivalent patient engagement with virtual treatment. Participant’s families also reported on unexpected benefits arising from this adaptation: “Families in our trial reported several benefits of virtual treatment, including reduced burden of a lengthy commute and expanded access to care. Telehealth also provided a unique opportunity to conduct behavioral exposures and home-based exercises in the natural context of the patients own home, which was more reflective of the youths’ day-to-day lives” (Shear et al., 2022).
The PeRSEVERE principles apply to research involving active, ongoing human participation and are useful guidance for protecting trial integrity and participant rights when changes occur throughout the trial. The principles take account of the complexity of participation and what should happen when participants stop, reduce or change their involvement. There may be multiple elements to taking part, which could change, including participants attending hospital and being assessed; taking medicines; completing questionnaires. The 17 principles can be viewed here.
Use a “risk of bias” tool, before the trial to identify and address areas of risk and anticipated change (e.g. ROBUST-RCT; see also the Cochrane Risk of Bias resources)
How funders can ensure this happens
- Point research teams to the above resources.
- Ask research teams about protocol amendments that will take account of future adaptability and technologies used in their trial and how these might be anticipated to shift during the trial’s lifecycle.
- Ask research teams to describe their mechanisms for assessing this, and the potential for adapting the trial protocol to maintain scientific and ethical relevance.
- An example question to ask is given in the INFORM Structured Grant Form for Trials, Question {27}.