Good Practice Action 3: Working with patients and communities
Engage patients and communities from the outset to build trust, ensure relevance, and strengthen recruitment and retention
Engagement that begins early and is grounded in local context helps ensure trial questions reflect real priorities of importance to patients and communities, interventions are acceptable, and participation is sustained through trust and shared ownership.
Watch the video: INFORM Good Practice Action 3
Clarinda Cerejo, Patient Advocate and EUPATI Fellow explains why engaging with patients and communities early in the design of a trial is essential for trial informativeness.
Trial teams and others should:
Through these interactions, trial hypotheses can be carefully set to address real priorities, and proposed interventions can be evaluated to ensure they are acceptable from the perspective of those with lived experience. Building trusted relationships is important to foster effective recruitment and retention strategies. Without the right participants, trials may fail to address priorities that are relevant to patients and communities or may implement interventions that are not acceptable or feasible from their perspective. This can lead to challenges in recruitment, retention, and overall trial success.
Research teams should engage with patients and communities for collaborative partnerships that are grounded in local context from the earliest stage possible.
A “community” may refer to populations connected to or affected by the trial, which may be defined geographically or through shared conditions or social contexts, and whose local knowledge can help ensure trial processes are relevant, effective and acceptable.
Patient- and participant-centricity are lenses through which to conceptualise, design and conduct informative trials. These perspectives bring attention also to the practical, day-to-day factors that influence whether individuals can participate in research and are particularly important for under-served or marginalised groups, for whom logistical, financial, and physical burdens can significantly limit access. By anticipating these challenges and embedding appropriate solutions into trial protocols, research teams can improve inclusivity, participant experience, and study feasibility. For resources to support the design of inclusive trials, see Inclusive Trials Resources.
The principles of research co-production are helpful when thinking about building trusted partnerships:
- avoiding tokenism
- including listening to all perspectives
- respecting and valuing all input
- reciprocity
- balanced power dynamic
Read more on co-production in health research in the blog post from UCLPartners Health Innovation.
Research teams planning clinical trials can refer to helpful guidance for engaging communities and patient and public involvement (PPI) in an inclusive way. For example, the work published by Anderson and colleagues describes a knowledge mobilisation approach to build trust and inclusion through community engagement (Anderson et al., 2024).
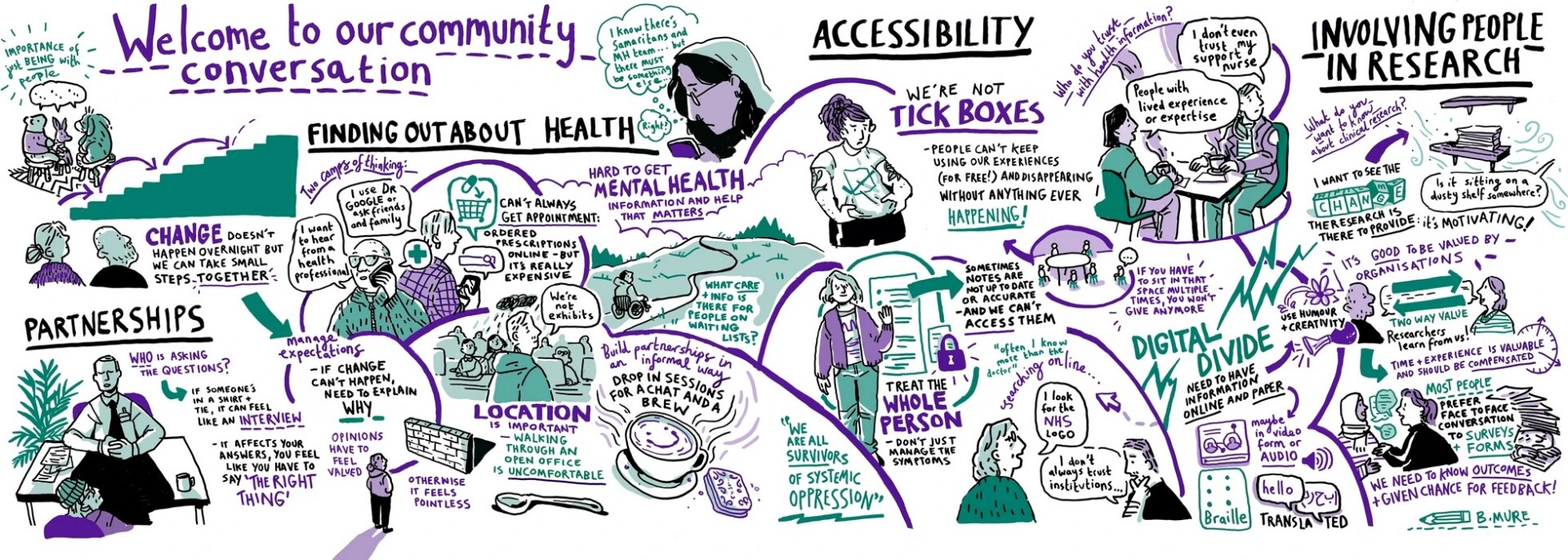
The Figure below illustrates the many components that make a difference to building good quality partnerships with communities, based on trust and inclusion.
Figure 1. From Anderson et al. 2024. Visual notes illustrated from expert citizens community conversations. Licenced by CC BY 4.0.
Research teams can refer to the table of 38 co-produced lessons learned for future engagement with community organisations, from Anderson et al., 2024.
The STRIDE recommendations remind us that these discussions need time, and trust-building is a long-term commitment.
A ‘Checklist for inclusive community involvement in health research’ (CHICO) guidance (NIHR Bristol BRC, 2023): sets out three areas for researchers to think about when building relationships with communities:
- Building relationships – Building trust with the community, rather than researchers ‘parachuting’ in to gain input to a project without returning
- Reciprocal relationships – An ongoing and iterative process to understand the best ways to build and maintain trusting and mutually beneficial relationships
- Practicalities – Developing and running inclusive community group involvement activities need careful thought to the practicalities of doing so. Really understanding what would work best for a particular community group is important here
What this means for researchers is set out in the CHICO guidance table.
Building partnerships needs time, along with effective and appropriate communication methods. Embedding patient and community members in advisory boards, and PPI groups for input to trial protocols creates an enduring source of valuable contribution.
How funders can ensure this happens
- Funders should point research teams to helpful guidance such as that set out above.
- Ask about their plans for how patients, service users and public contributors have been and will continue to be involved in the trial design.
- Ask about their recruitment and retention strategies, and how they have discussed and tested these strategies with PPI and relevant communities.
- Example questions to ask are in the INFORM Structured Grant Form for Trials, Questions {6, 18, 20 and 21}