Good Practice Action 11: Ensure trial registration and results published
Ensure trial registration is monitored, and results are published in a timely and complete manner to maintain accurate records, meet ethical obligations, and reduce selective reporting and publication bias
Active monitoring of trial registration and reporting promotes transparency, protects research integrity, and ensures that all findings, regardless of outcome, contribute to the broader evidence base. Failure to share and learn from all research, including research that does not produce a result in favour of the treatment under test, has been recognised as a key source of clinical trial research waste and inefficiency. Selective or lack of reporting prevents the opportunity to build on previous research and can lead to duplication of effort.
Watch the video: INFORM Good Practice Action 11 [To come]
Trial teams and others should:
Research teams should use Open Science (OS) practices and trial registration (see Good Practice Action 4).
Funding that is conditional on a mandate that trials are registered prospectively, and that there is a system for monitoring the upkeep of records and reporting upholds accountability and transparency in trials. This ensures that all findings are published and shared within a relevant timeframe so that lack of publication or selective interpretation of results do not add to research waste (Figure 1). There should be a plan for sharing anonymous participant data, including the data dictionary, statistical code and other relevant materials to enable others to examine then data behind the trial results.
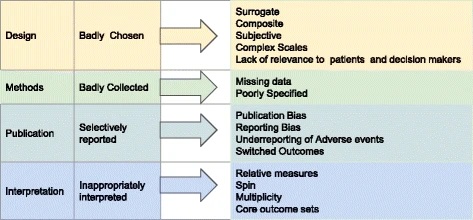
Figure 1: Why clinical trial outcomes fail to translate into benefits for patients, from Heneghan et al., 2017 licenced by CC BY 4.0.
Researchers can work towards creating a culture of learning in response to evidence that an intervention was ineffective or harmful, both for the intervention itself and the wider field of clinical trials (Axford et al., 2020). Every result adds valuable insight to the evidence base.
The Sense about Science AllTrials campaign estimated that in 2013 over half of all clinical trial results were never reported. When results are cherry-picked, the evidence base is distorted. The AllTrials campaign therefore aims to have every trial registered and reported. EU Trials Tracker is a searchable database updated monthly to gather information on trials in the EU since 2004. This tracks which universities and pharmaceutical companies are registering and reporting their trials (and those which are not).
How funders can ensure this happens
- Funders can encourage, or mandate, research teams to use OS practices.
- Ask research teams to name the trial registry they will use to register the trial (this should be on the WHO International Clinical Trials Registry Platform or clinicaltrials.gov).
- Funders can mandate that trial registration is a condition of funding and will be checked at month [x] as part of the funding agreement.
- Request that the trial findings are published in full, in an accessible form within [x] months of the funding end date, and that the results are added to the trial registry. This should include all findings, regardless of outcome and not a selection of the findings.
- Ask how trial teams plan to share the participant data, including the data dictionary, statistical code and other relevant materials to enable others to examine the data behind the trial results.
- Example questions to ask are given in the INFORM Structured Grant Form for Trials, Questions {31, 32 and 33}
