Good Practice Action 4: Use existing design tools
Apply structured design tools, established frameworks, and Open Science practices to improve trial quality, transparency, and reproducibility
Using structured tools and frameworks and aligning to Open Science (OS) practices embeds consistency and accountability across the trial lifecycle and supports comprehensive and coordinated design, conduct, and reporting.
Watch the video: INFORM Good Practice Action 4
Alejandra Recio Saucedo, Senior Research Fellow, University of Southampton, UK, explains why using existing design tools and open research processes are essential for an informative trial.
“The moment a research idea is born is the perfect moment for us to search registries. This is the time when innovation happens and it is also where we can find collaborators. Overall discovery of what has been done before through registries and pre-registration allows us to incorporate existing findings into newer, more focused designs.”
Trial teams and others should:
Research teams should use existing tools, frameworks and guides from the earliest stage and throughout the trial lifecycle to maximise the quality of health research. The accountability and transparency of trials should also be upheld by pre-registering the trial or submitting registered reports (see Good Practice Action 11).
Without this, there may be design flaws, gaps, inconsistencies and uncertainties, deeming the findings of limited use, or the trial entirely futile, contributing to research waste.
Tools, framework and guides | Description | |
Core outcome sets (COS) are an agreed standard set of outcomes that should be measured and reported, as a minimum, in all clinical trials in specific areas of health or healthcare to allow harmonised reporting. The COMET Initiative brings together people interested in developing and application of COS. | Video explaining how PPI can input on COS COMET’s searchable database of studies where COS have been described for a particular disease of condition | |
A tool to visualise trial elements according to how pragmatic (trials that are true to real-world conditions) versus how explanatory (trials conducted under very narrow conditions) these elements are | Video explaining the purpose and use of PRECIS-2 | |
Standard Protocol Items: Recommendations for Interventional Trials, a checklist of items to include in a trial protocol. This includes updated prompts for researchers to include information about their OS practices | Updated SPIRIT section on OS practices | |
A tool developed following on from PROGRESS-Plus (Cochrane, 2025) which recommends six core participant characteristics (sex, gender, ethnicity, age, socioeconomic status, and location) to include in every trial, and additional characteristics to consider, depending on your trial | Video explaining the purpose and use of PRO EDI | |
CONsolidated Standards Of Reporting Trials – a checklist of items to include in trial reporting. The same checklist is useful to inform the trial’s design |
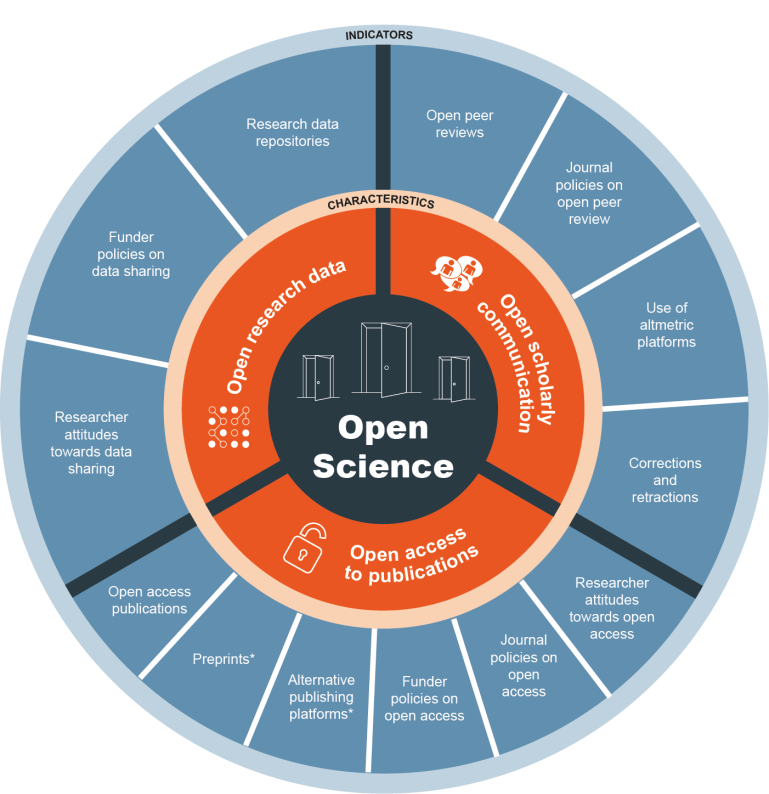
OS practices are based on principles of inclusion, equity, and transparency to improve rigour and accountability and to make research more reliable and bring greater benefits in an equitable way (Figure 1).
Figure 1. The characteristics and indicators of Open Science, Open Access Belgium, 2025 licenced by CC BY 4.0.
How funders can ensure this happens
- Funders should point research teams to existing tools, frameworks and guides (such as those shown above) in their pre-submission process.
- Ask research teams to name the trial registry they will use to register the trial (this should be on the WHO International Clinical Trials Registry Platform or clinicaltrials.gov).
- Funders can mandate that trial registration is a condition of funding and will be checked at month [x] as part of the funding agreement.
- Example questions to ask are given in the INFORM Structured Grant Form for Trials, Questions {2, 14, 15 and 31}