Good Practice Action 10: Have external oversight
Maintain oversight throughout the lifecycle of a trial through ethics review boards, trial steering committees, and data monitoring to uphold scientific rigour and support evidence-informed analyses
Effective oversight ensures that ethical standards are upheld, emerging risks are managed, and analyses are guided by credible evidence, maintaining confidence in both the trial process and its outcomes. This is key for having transparency and rigour in the research. Without this, there are ethical and integrity risks to the trial
Watch the video: INFORM Good Practice Action 10 [To come]
Trial teams and others should:
Having oversight from ethics review boards, trial steering committees, and a committee for data protection and monitoring can foster better quality trials with higher chances of being successful and informative.
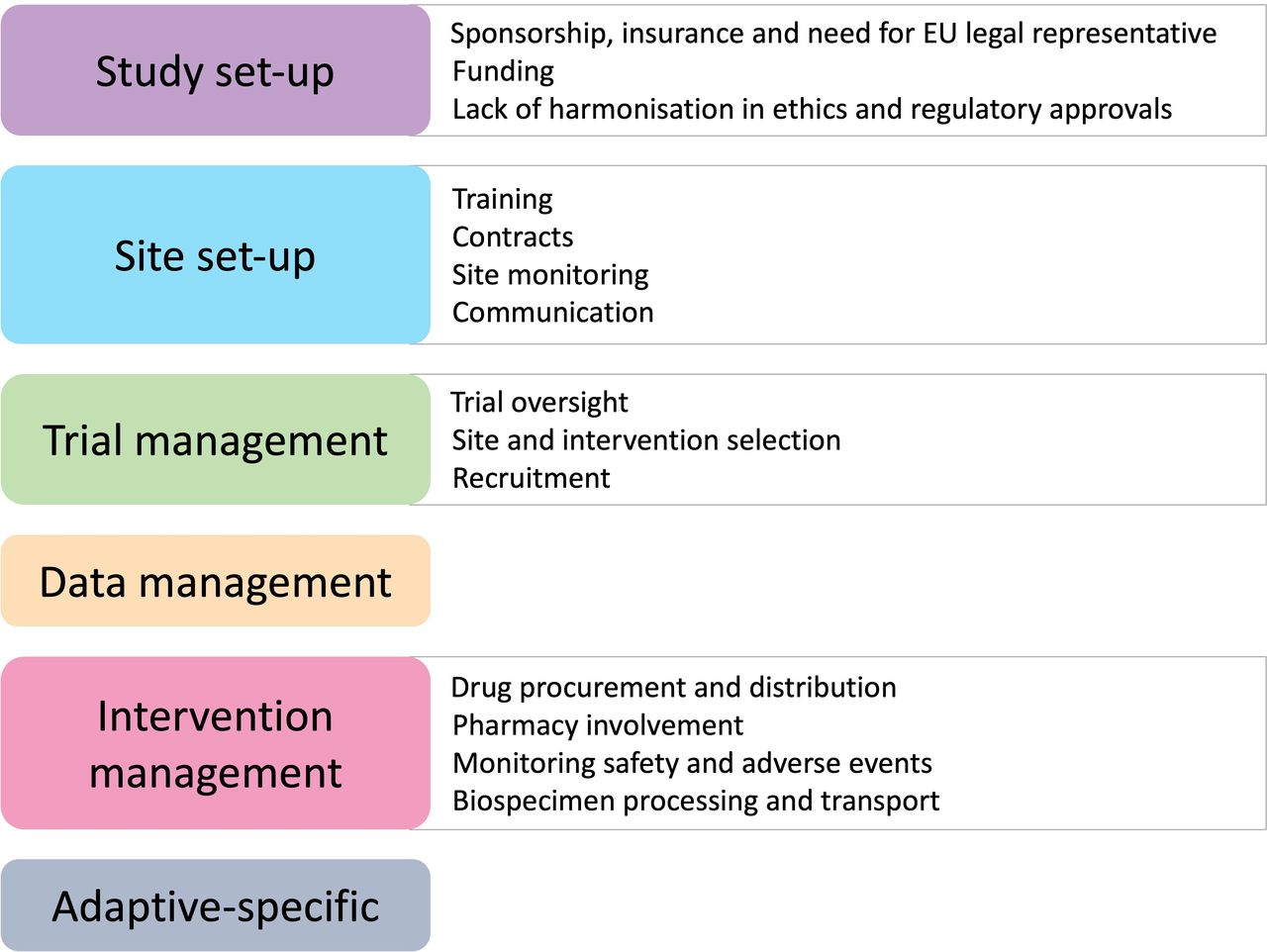
International trials face complex operational challenges from funding and obtaining regulatory approvals, to site contracts and drug distribution (summarised in Figure 1). Careful planning and communication with sites and key stakeholders during the trial planning phase is essential (Gumber et al., 2024).
Figure 1: Operational complexities of international trials, from Gumber et al., 2024 licenced by CC BY 4.0.
Where multi-site trials operate across jurisdictions, trial teams should aim to harmonise ethics submissions where possible to avoid a duplication of effort.
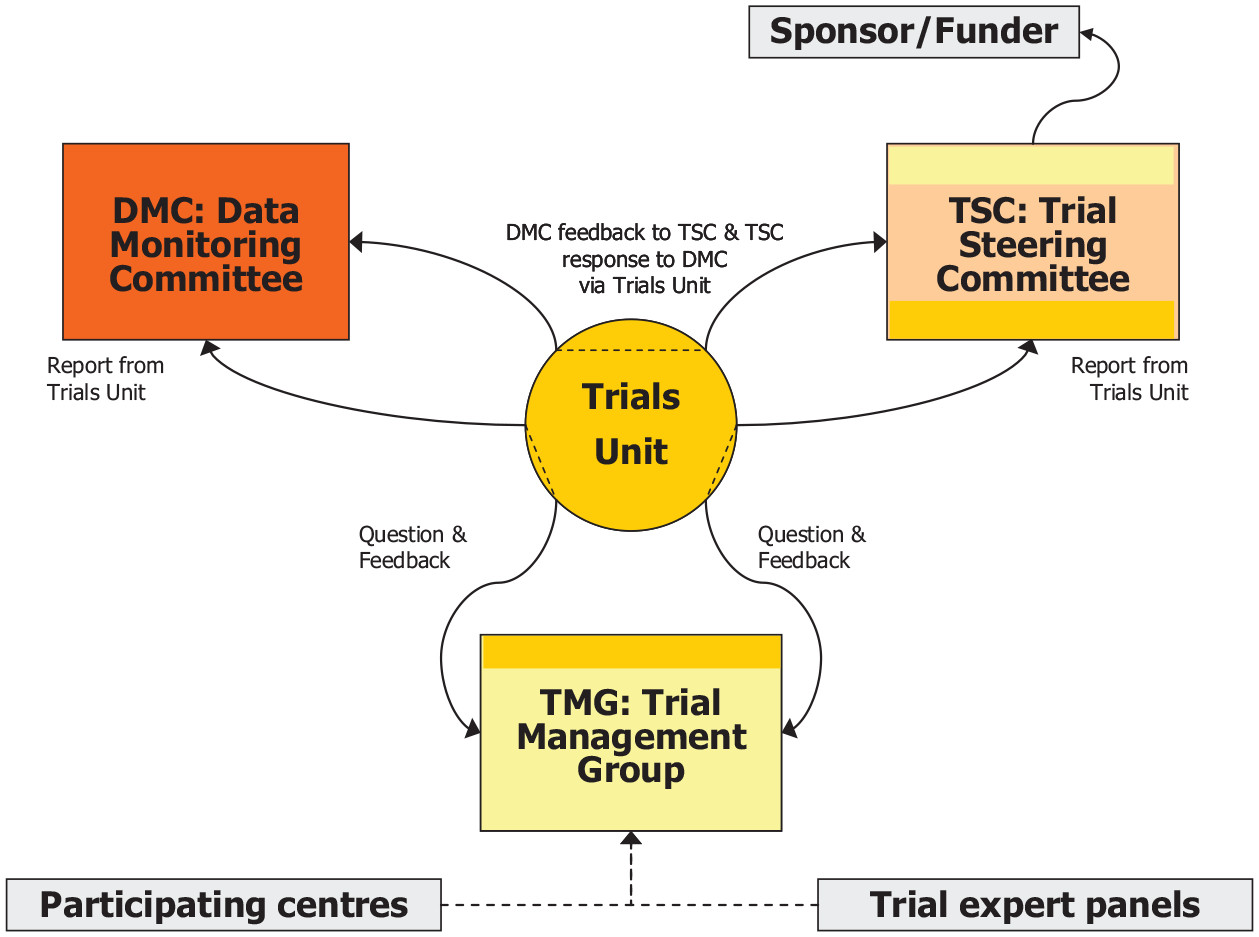
Oversight can be achieved from the input of committees with differing structure, independence and responsibilities (Lane et al., 2020), shown in Figure 2.
Figure 2. Oversight committee structures, from Lane et al., 2020, licenced by CC BY 4.0.
A trial steering committee can oversee that the trial is conducted in accordance with legislation, regulations, guidelines, and best practices and can advise on, and can take action if there are potential research violations. Upholding the participants’ rights, safety and wellbeing are central to a trial steering committees remit (NIHR, 2023). While having holistic oversight of the trial to represent all who could be impacted (participants, patients, trialists, funder, sponsor) currently and in the future was noted by an expert panel discussing the function of trial steering committees (Harman et al., 2015).
A data protection committee can ensure compliance with relevant data protection laws and best practices to ensure robust data handling. Multi-site trials may have different data considerations across jurisdictions, or unique contextual factors such as the use of Indigenous data and knowledge. The DAta Monitoring Committees: Lessons, Ethics, Statistics (DAMOCLES) Study Group have proposed a data protection committee template (Campbell et al., 2005)
An example is given from the British Heart Foundation of proposed requirements for trial steering committee and data protection committee.
How funders can ensure this happens
- Ask research teams about their plan for obtaining research ethics committee/institutional review board (REC/IRB) and other approvals.
- Ask research teams to provide a plan for the external oversight of their trial. These are likely to include a Steering Committee and a Data Monitoring and Ethics Committee but there may be others depending on the nature of the trial.
- Example questions to ask are given in the INFORM Structured Grant Form for Trials, Questions {26 and 29}